Unit 2: Atoms & the Periodic Table Standards
SPS1. Obtain, evaluate, and communicate information from the Periodic Table to explain the relative properties of elements based on patterns of atomic structure.
a. Develop and use models to compare and contrast the structure of atoms, ions and isotopes. (Clarification statement: Properties include atomic number, atomic mass and the location and charge of subatomic particles.)
b. Analyze and interpret data to determine trends of the following:
• number of valence electrons
• types of ions formed by main group elements
• location and properties of metals, nonmetals, and metalloids
• phases at room temperature
c. Use the Periodic Table as a model to predict the above properties of main group elements.
a. Develop and use models to compare and contrast the structure of atoms, ions and isotopes. (Clarification statement: Properties include atomic number, atomic mass and the location and charge of subatomic particles.)
b. Analyze and interpret data to determine trends of the following:
• number of valence electrons
• types of ions formed by main group elements
• location and properties of metals, nonmetals, and metalloids
• phases at room temperature
c. Use the Periodic Table as a model to predict the above properties of main group elements.
Topics:
Students are expected to know and learn:
- Structure of an atom
- Atomic number & mass
- Proton number and element identity
- Valence electrons
- Isotopes
- Types of ions
- Element phases at room temperature
- Organization of the Periodic Table
- Element properties based on Periodic Table
Reading Assignment: Book Chapters

Chapter 17: Properties of Atoms and the Periodic Table
Sections 1, 2, & 3
Chapter 19: Elements and Their Properties
Section 1, 2, &3
Sections 1, 2, & 3
Chapter 19: Elements and Their Properties
Section 1, 2, &3
Atomic Structure
Essential Question: How do I develop and use models to compare and contrast the structure of atoms, ions, and isotopes? (In terms of atomic number, atomic mass, and the location and charge of subatomic particles.)
Access the SlideShare presentation by clicking HERE!
Access the SlideShare presentation by clicking HERE!
Neutral Atoms, Isotopes, and Ions
Watch the following video for more information to compare and contrast atoms, isotopes, and ions.
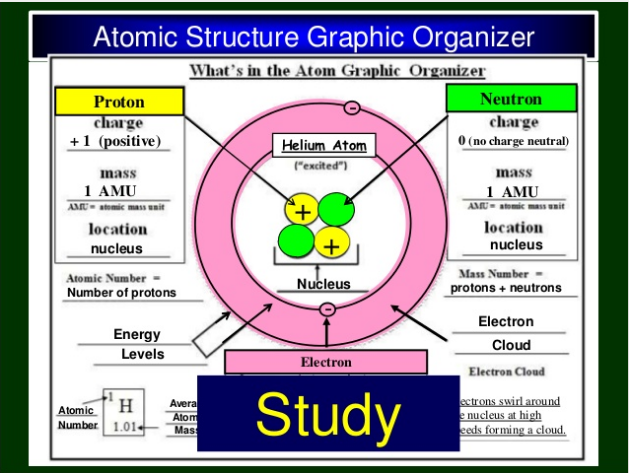
The following Graphic Organizer explains how atoms compare based on the subatomic particles. You must understand the similarities and differences between neutral atoms, ions, and isotopes.
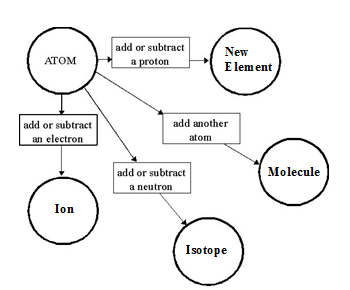
Neutral Atom: A neutral atom (of any element) has an equal number of protons and electrons. If you change the number of protons it will no longer be the same element. Protons identify the element, and are the same as the atomic number.
Molecule: A molecule is a chemical bond of two or more atoms of the same element. Ex: H2
Isotope: Atoms of the same element can have a different number of neutrons. Therefore, isotopes of the same element have a different relative atomic mass. The atomic mass identified on the Periodic Table is the average atomic mass of all the isotopes that exist for that element.
Ion: Atoms of an element may gain or lose electrons. This causes the total number of protons and electrons to be different (unequal). If an atom gains electrons, it becomes an ANION (a negative ion). If an atom loses electrons, it becomes a CATION (a positive ion). Ions are created by chemical means when an atom bonds with another atom.
Molecule: A molecule is a chemical bond of two or more atoms of the same element. Ex: H2
Isotope: Atoms of the same element can have a different number of neutrons. Therefore, isotopes of the same element have a different relative atomic mass. The atomic mass identified on the Periodic Table is the average atomic mass of all the isotopes that exist for that element.
Ion: Atoms of an element may gain or lose electrons. This causes the total number of protons and electrons to be different (unequal). If an atom gains electrons, it becomes an ANION (a negative ion). If an atom loses electrons, it becomes a CATION (a positive ion). Ions are created by chemical means when an atom bonds with another atom.
Isotopes
Click on the Isotope button below to read more about Isotopes. It will explain how a neutral atom becomes an Isotope. Watch the video at the end and answer the following questions.
Click on the Isotope button below to read more about Isotopes. It will explain how a neutral atom becomes an Isotope. Watch the video at the end and answer the following questions.
Ions
Click on the Ion button below to read more about Ions. It will explain how a neutral atom becomes an Ion. Watch the video at the end and answer the following questions.
Click on the Ion button below to read more about Ions. It will explain how a neutral atom becomes an Ion. Watch the video at the end and answer the following questions.
Build an Atom PhET Simulation
Use the Ions Lab Activity Handout to complete the Build an Atom PhET Simulation.
Atoms, Ions, & Isotopes Quizlet
Review the atoms, ions, and isotopes quizlet before the quiz. :)
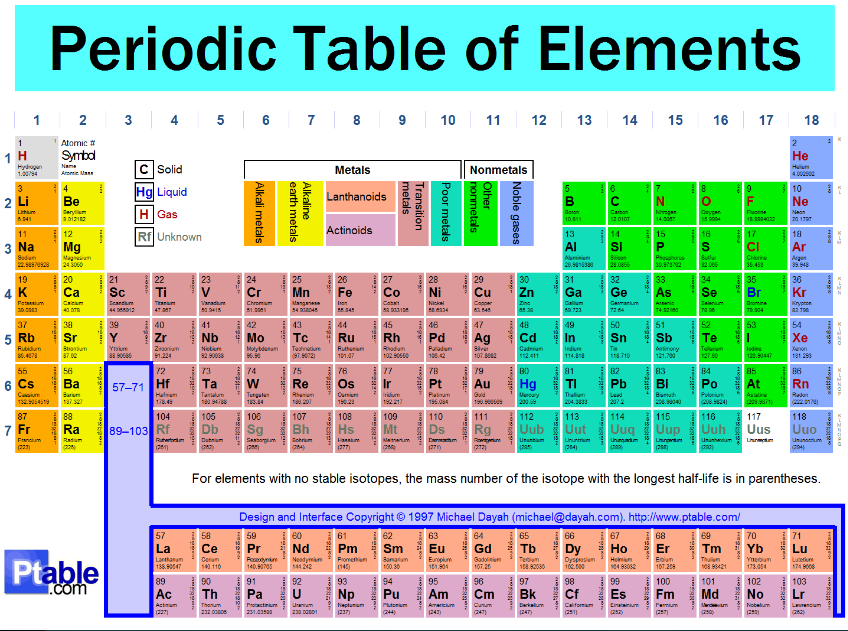
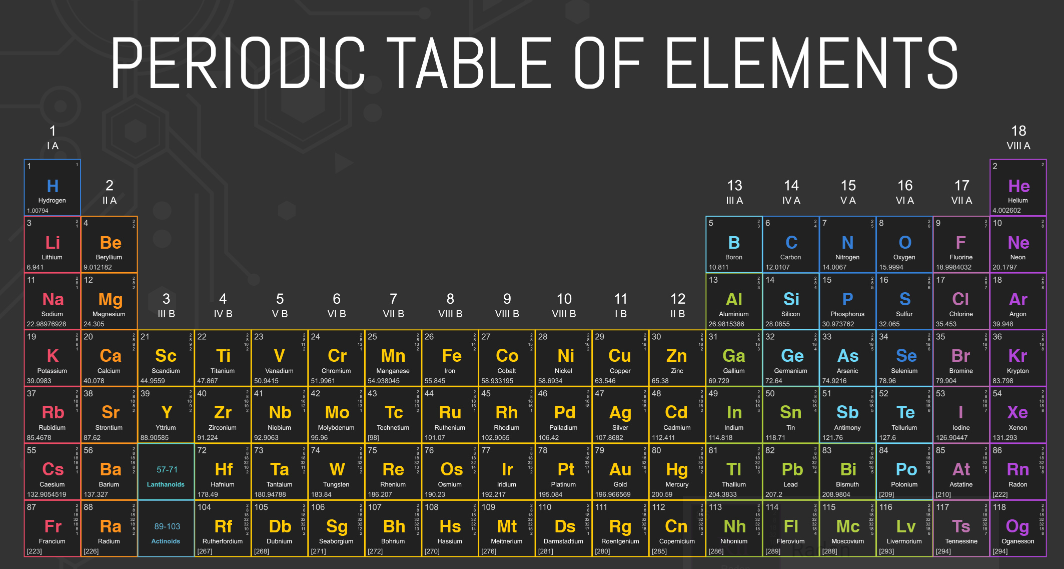
The Periodic Table
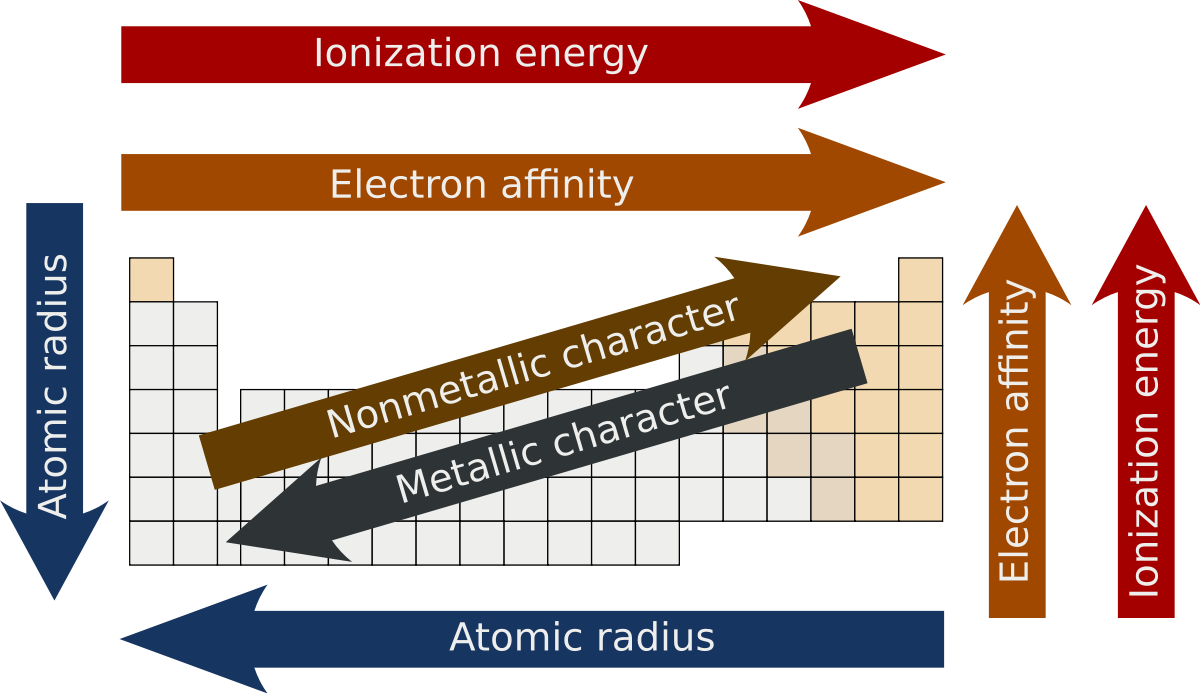
Essential Question: How do I analyze and interpret data to determine trends of the following: number of valence electrons, types of ions formed by main group elements, location of metals, nonmetals, and metalloids, and phases at room temperature?
Access the Dynamic Periodic Table by clicking HERE!!!
Access the Dynamic Periodic Table by clicking HERE!!!
Trends of the Periodic Table
Video- The Periodic Table, Crash Course Chemistry
Periodic Table Quizlet Review
Review the Periodic Table Quizlet below using your labeled periodic table before the Quiz. :)
Periodic Table of Tech
Click the link to visit the Periodic Table of Tech website to learn how different elements are used in Technology!