Unit 1 Matter: Standards
SPS5. Obtain, evaluate, and communicate information to compare and contrast the phases of matter as they relate to atomic and molecular motion.
a. Ask questions to compare and contrast models depicting the particle arrangement and motion in solids, liquids, gases, and plasmas.
b. Plan and carry out investigations to identify the relationships among temperature, pressure, volume, and density of gases in closed systems. (Clarification statement: Using specific Gas laws to perform calculations is beyond the scope of this standard; emphasis should focus on the conceptual understanding of the behavior of gases rather than calculations.)
SPS7. Obtain, evaluate, and communicate information to explain transformations and flow of energy within a system.
d. Analyze and interpret data to explain the flow of energy during phase changes using heating/cooling curves.
a. Ask questions to compare and contrast models depicting the particle arrangement and motion in solids, liquids, gases, and plasmas.
b. Plan and carry out investigations to identify the relationships among temperature, pressure, volume, and density of gases in closed systems. (Clarification statement: Using specific Gas laws to perform calculations is beyond the scope of this standard; emphasis should focus on the conceptual understanding of the behavior of gases rather than calculations.)
SPS7. Obtain, evaluate, and communicate information to explain transformations and flow of energy within a system.
d. Analyze and interpret data to explain the flow of energy during phase changes using heating/cooling curves.
Topics:
Students are expected to know and learn:
- Chemical and physical properties
- Compare/Contrast states of matter
- Relationship between temperature, pressure, volume and density (Boyle's Law and Charles's Law)
- Classifying matter
- Flow of energy through a phase change diagram
Reading Assignment: Book Chapters

Chapter 15: Classification of Matter
Sections 1 & 2
Chapter 16: Solids, Liquids, and Gases
Sections 1 & 3
Sections 1 & 2
Chapter 16: Solids, Liquids, and Gases
Sections 1 & 3
|
Video
Article: Scientists find new state of matter which is both solid and liquid at the same time
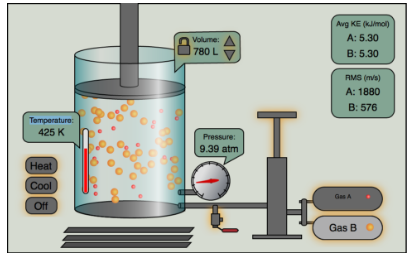
Pressure, Temperature, and Volume
Essential Question: How do I plan and carry out investigations to identify the relationship among temperature, pressure, volume and density of gases in closed systems?
Activity: Complete the Kinetic Molecular Theory using the Chem 301- Gas Law Simulator. Follow the instructions on the handout provided. This assignment will be posted in Google Classroom.
Activity: Complete the Kinetic Molecular Theory using the Chem 301- Gas Law Simulator. Follow the instructions on the handout provided. This assignment will be posted in Google Classroom.
Your browser does not support viewing this document. Click here to download the document.
Video- Behavior of Gases
Three Station Gas Law Lab
Your browser does not support viewing this document. Click here to download the document.
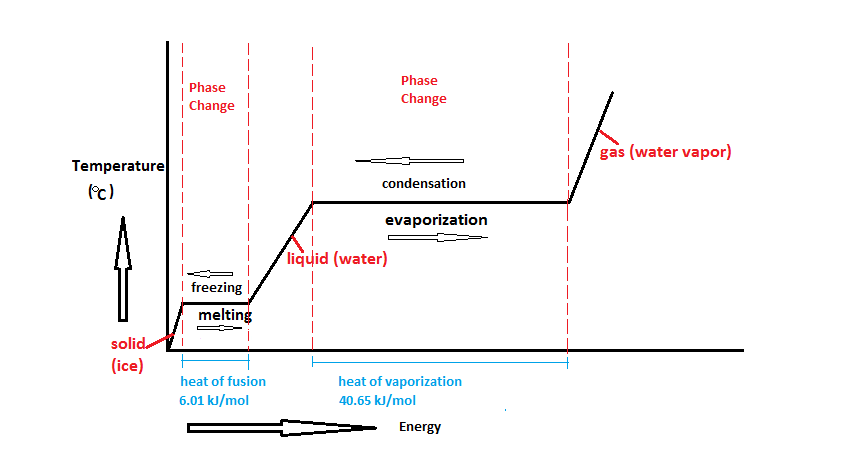
The Phase Change Diagram
SPS7. Obtain, evaluate, and communicate information to explain transformations and flow of energy within a system.
d. Analyze and interpret data to explain the flow of energy during phase changes using heating/cooling curves.
Watch the following video on Heating Curve Basics.
d. Analyze and interpret data to explain the flow of energy during phase changes using heating/cooling curves.
Watch the following video on Heating Curve Basics.
Notice that, in general, the temperature goes up the longer the heating continues. However, there are two horizontal flat parts to the graph. These happen when there is a change of state. The plateaus are also called phase changes.
The first change of state is melting (changing from a solid to a liquid). The temperature stays the same while a substance melts. For water, this temperature is 0°C because the melting point for water is 0°C.
The second change of state is boiling (changing from a liquid to a gas). The temperature stays the same while a substance boils. For water, this temperature is 100°C because the boiling point for water is 100°C.
Different substances have different melting points and boiling points, but the shapes of their heating curves are very similar.
The first change of state is melting (changing from a solid to a liquid). The temperature stays the same while a substance melts. For water, this temperature is 0°C because the melting point for water is 0°C.
The second change of state is boiling (changing from a liquid to a gas). The temperature stays the same while a substance boils. For water, this temperature is 100°C because the boiling point for water is 100°C.
Different substances have different melting points and boiling points, but the shapes of their heating curves are very similar.
Activity: Summarize the flow of energy (change in thermal energy and temperature) as you move up the heating curve and then move down the cooling curve. Be specific as to tell what occurs during the phase and in between phase change. After you finish, answer the following questions from Quizlet: Phase Change Diagram.