Energy & Heat Standards
SPS7. Obtain, evaluate, and communicate information to explain transformations and flow of energy within a system.
a. Construct explanations for energy transformations within a system. (Clarification statement: Types of energy to be addressed include chemical, mechanical, electromagnetic, light, sound, thermal, electrical, and nuclear.)
b. Plan and carry out investigations to describe how molecular motion relates to thermal energy changes in terms of conduction, convection, and radiation.
c. Analyze and interpret specific heat data to justify the selection of a material for a practical application (e.g., insulators and cooking vessels).
d. Analyze and interpret data to explain the flow of energy during phase changes using heating/cooling curves.
a. Construct explanations for energy transformations within a system. (Clarification statement: Types of energy to be addressed include chemical, mechanical, electromagnetic, light, sound, thermal, electrical, and nuclear.)
b. Plan and carry out investigations to describe how molecular motion relates to thermal energy changes in terms of conduction, convection, and radiation.
c. Analyze and interpret specific heat data to justify the selection of a material for a practical application (e.g., insulators and cooking vessels).
d. Analyze and interpret data to explain the flow of energy during phase changes using heating/cooling curves.
Reading Assignment: Book Chapters

Chapter 4: Energy
Sections 1, 2, & 3
Chapter 6: Thermal Energy
Sections 1 & 2
Sections 1, 2, & 3
Chapter 6: Thermal Energy
Sections 1 & 2
Phenomenia: How a Roller Coaster functions...
Unit 6 Powerpoint Notes
Energy Transformations
a. Construct explanations for energy transformations within a system. (Clarification statement: Types of energy to be addressed include chemical, mechanical, electromagnetic, light, sound, thermal, electrical, and nuclear.)
Watch the following video in order to construct an explanation for energy transformations within a system. The video will discuss how to calculate efficiency. This is not part of the standard.
Guiding Questions:
What is the difference between stored energy(potential) and energy in motion(kinetic)?
Is energy input equal to energy output? Explain what happens.
Draw a SANKEY Diagram and explain how it relates to energy transformation.
Watch the following video in order to construct an explanation for energy transformations within a system. The video will discuss how to calculate efficiency. This is not part of the standard.
Guiding Questions:
What is the difference between stored energy(potential) and energy in motion(kinetic)?
Is energy input equal to energy output? Explain what happens.
Draw a SANKEY Diagram and explain how it relates to energy transformation.
Energy Forms & Changes pHet Simulation
Use the following phET simulation to complete the Energy Forms & Changes handout. Click on Systems below to begin!
Phenomena: Let's Learn S'more about Heat!
Why does a marshmallow get fat and squishy when you heat it in a microwave or convection oven, but will change into the ideal toasted marshmallow when cooked on an open flame?
Have you ever roasted a marshmallow over a campfire? Have you smelt the smoke coming from the roasted marshmallow? Or did you feel warmth from the fire? In each of these situations, you experienced the work of the three types of heat transfer: conduction, convection, and radiation. Conduction, or direct heat, occurs when the faster moving molecules of the fire collide with the cooler molecules of the marshmallow, causing them to move faster and become hot.
Diagram 1
Have you ever roasted a marshmallow over a campfire? Have you smelt the smoke coming from the roasted marshmallow? Or did you feel warmth from the fire? In each of these situations, you experienced the work of the three types of heat transfer: conduction, convection, and radiation. Conduction, or direct heat, occurs when the faster moving molecules of the fire collide with the cooler molecules of the marshmallow, causing them to move faster and become hot.
Diagram 1

The diagram (#1) to the right shows the metal rod conducting heat to the person's hand. Some solids, unlike the metal rod, are poor conductors of heat, making them excellent for insulating heat. These solids are called insulators. Some examples of insulators would include wood or plastic, which is often used for making handles for pots and pans. Conduction is also found in movie theaters. Popcorn machines used radiation (another type of heat transfer), and convection. Convection is heat transfer by air, or liquid. the popcorn is heated by the air surrounding it. The light inside the popcorn machine produces infrared rays, which proves that popcorn machine produces radiation. You may not know it, but just the scent of popcorn is a form of convection. Back home, let us say that you are cooking something on the stove. This would be a form of conduction. The hot molecules from the stove collide with the cool molecules of the food. This is called conduction or direct heat.
Diagram 2
Diagram 2

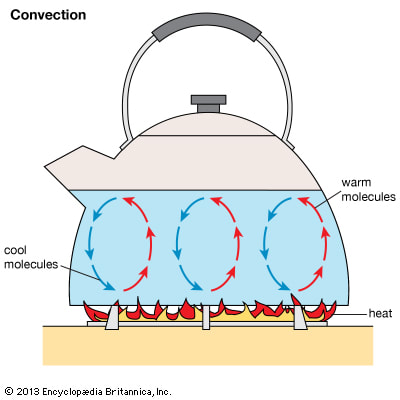
The picture to the left (diagram #2) shows a thermos, another example of an insulator. People use the thermos for keeping water the same temperature it originally was for a considerable amount of time. The thermos creates a vacuum which keeps the speed of the molecules the same. Instead letting the warm and cool molecules collide, the thermos doesn't let any air in or out. When heat is transferred through liquids such as water, or gases, this is called convection. Typical examples include breathing hot air on your hands during a cold day. The heat from your mouth warms up your hands. This transfer of heat by air is convection.
When liquids such as water are heated, their molecules move faster and farther apart. This decreases their density and makes the molecules lighter. Now, the heavier surrounding molecules sin down, pushing the lighter molecules upward, creating a convection current. One may have never realized this, yet there are convection currents everywhere. Convection currents tell us how wind currents are formed, how houses are heated, how birds can fly, and even why smoke rises.
Diagram 3
Diagram 3
Diagram 3, above shows how warm air rises and cool air sinks in the air. This is so, since the heated warm molecules are pushed upwards by the heavier cool air molecules. This is why warm air rises and cool air sinks. This doesn't happen so just in the air, but in our homes as well. But why does hot air rise? This is because of the Principle of Buoyancy, where denser molecules sink, and the less dense molecules, such as the hot air molecules, rise. Take a cork, for example. If one would drop the cork into a jar of water, it would float, since it is less dense. Water has a density of 1. Therefore, the cork has a density less than one since it floats. What would happen if you dropped a coin in to the jar? It would sink as it is denser. The Principle of Buoyancy does not only apply to liquids, but to gasses as well.
Unlike conduction or convection, radiative heat energy can be transferred through a vacuum in the form of electromagnetic waves by radiation. This is similar to the way light and radio waves travel. These heat waves, also known as infrared waves, travel from heat sources such as the sun, a light bulb, or even a flashlight!
Unlike conduction or convection, radiative heat energy can be transferred through a vacuum in the form of electromagnetic waves by radiation. This is similar to the way light and radio waves travel. These heat waves, also known as infrared waves, travel from heat sources such as the sun, a light bulb, or even a flashlight!

Diagram 4 (right) shows a great example of radiation playing a role in our daily lives. Ninety-three million miles away, the sun sends out infrared heat waves through space, to the Earth. The rays enter the atmosphere, and soon reach the clouds, where the rays reflect off the clouds. Not all of the infrared rays are reflected off the clouds; some are absorbed by water, dust, and 03. While infrared rays are being emitted by the sun, infrared radiation is being emitted by the surface of the Earth. These rays are being sent through sky, out of the atmosphere, and into space. Radiation not only occurs in outer space, but also in our every day lives. Infrared waves can be sent through light bulbs. There are several different types of light bulbs- incandescent, flourescent, halogen, LED. Incandescent bulbs produce 98% heat energy. If you touch an incandescent light bulb, you would experience conduction. If you keep your hands close to the bulb, you would feel warm air. This is convection.
Congratulations! You have learnt all about the three types of heat transfer!
Congratulations! You have learnt all about the three types of heat transfer!
Video: The Physics of Heat
Phenomena: Sand vs. Water at the Beach
What is specific heat? Why should we consider an object's specific heat capacity when determining it's application?
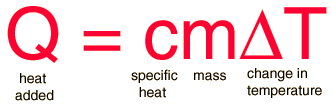
Specific heat is the heat required to raise the temperature of the unit mass of a given substance by a given amount (usually one degree). The relationship between heat and temperature change is usually expressed in the form shown below where c is the specific heat. The relationship does not apply if a phase change is encountered, because the heat added or removed during a phase change does not change the temperature.
The specific heat of water is 1 calorie/gram °C = 4.186 joule/gram °C which is higher than any other common substance. As a result, water plays a very important role in temperature regulation. The specific heat per gram for water is much higher than that for a metal, as described in the water-metal example.
The specific heat of water is 1 calorie/gram °C = 4.186 joule/gram °C which is higher than any other common substance. As a result, water plays a very important role in temperature regulation. The specific heat per gram for water is much higher than that for a metal, as described in the water-metal example.
Applications of Specific Heat Capacity
Why choose a pot, such as the one above, to cook your food in?
1. Different part of a cooking pot are made up of different material.
2. The base of a cooking pot is usually made up of copper because
a. copper has low specific heat capacity so that it need less heat to raise up the temperature.
b. copper is a good heat conductor.
c. copper has high density. The heavier base can make the pot become more stable.
3. The handles of cooking pot is usually made up of plastic or wood because
a. plastic and wood have high specific heat capacity. Their temperature won’t become too high even it absorbs large amount of heat.
b. plastic and wood are good heat insulator.
c. plastic and wood have low density hence they do not add much to the total weight of the pot.
4. The body of the cooking pot is usually made up of stainless steel because
a. steel has low specific heat capacity and hence need need less heat to raise up the temperature.
b. steel does not react chemically with the food.
1. Different part of a cooking pot are made up of different material.
2. The base of a cooking pot is usually made up of copper because
a. copper has low specific heat capacity so that it need less heat to raise up the temperature.
b. copper is a good heat conductor.
c. copper has high density. The heavier base can make the pot become more stable.
3. The handles of cooking pot is usually made up of plastic or wood because
a. plastic and wood have high specific heat capacity. Their temperature won’t become too high even it absorbs large amount of heat.
b. plastic and wood are good heat insulator.
c. plastic and wood have low density hence they do not add much to the total weight of the pot.
4. The body of the cooking pot is usually made up of stainless steel because
a. steel has low specific heat capacity and hence need need less heat to raise up the temperature.
b. steel does not react chemically with the food.