Nuclear Chemistry Standards
SPS4. Obtain, evaluate, and communicate information to explain the changes in nuclear structure as a result of fission, fusion and radioactive decay.
a. Develop a model that illustrates how the nucleus changes as a result of fission and fusion.
b. Use mathematics and computational thinking to explain the process of half-life as it relates to radioactive decay. (Clarification statement: Limited to calculations that include whole half-lives.)
c. Construct arguments based on evidence about the applications, benefits, and problems of nuclear energy as an alternative energy source.
a. Develop a model that illustrates how the nucleus changes as a result of fission and fusion.
b. Use mathematics and computational thinking to explain the process of half-life as it relates to radioactive decay. (Clarification statement: Limited to calculations that include whole half-lives.)
c. Construct arguments based on evidence about the applications, benefits, and problems of nuclear energy as an alternative energy source.
Reading Assignment: Book Chapter

Chapter 18: Radioactivity and Nuclear Reactions
Sections 1, 2, & 4
Sections 1, 2, & 4
Nuclear Chemistry
The following video discusses all topics that will be covered during this unit.
Radioactivity vs. Radiation
Read the following article HERE!!!
Answer the questions below using the article. Click on File in the embedded Google Document below. Then select Make a Copy. Rename the copy deleting "Copy of" and adding your first and last name. This will give you the ability to edit the document and submit it in Google Classroom.
Alpha Decay, Beta Decay, and Gamma Radiation
Fission and Fusion
Watch the following video in order to compare and contrast Nuclear Fission & Nuclear Fusion. You must be able to develop a model that illustrates how the nucleus changes as a result of both processes.
Quizlet Review:
Use the Quizlet below to review major vocabulary words in reference to Fission & Fusion.
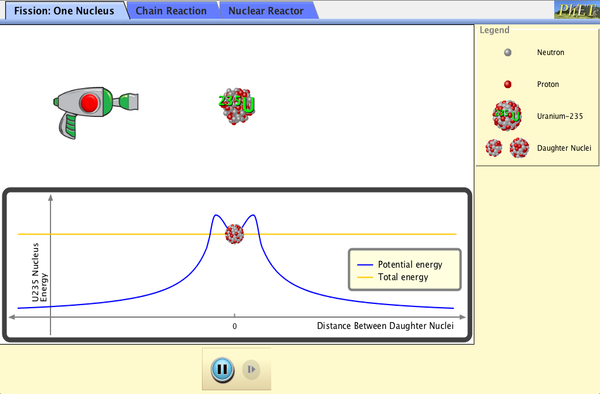
Fission: phET Simulation
The following phET simulation show the process of one nucleus undergoing a fission reaction once a neutron is fired at it. Change the screen to observe how a chain reaction results in many nuclei being split. Be sure to add the containment vessel to this activity. Finally, observe a picture of what a nuclear reactor looks like in a nuclear power plant. Fire neutrons into the reactor to see how power and energy are created. :)
Fusion: First U.S. Hydrogen Bomb Test
Think about how the results of the first U.S. Hydrogen Bomb is similar to a Nuclear Fusion reaction. Can you identify what the explosion looks like that is also an example of a Nuclear Fusion reaction?
Video: Most Radioactive Places on Earth
Do you have an idea of the most radioactive places on Earth? Watch the video until the end and be shocked at the TRUE most radioactive place on Earth! YIKES!
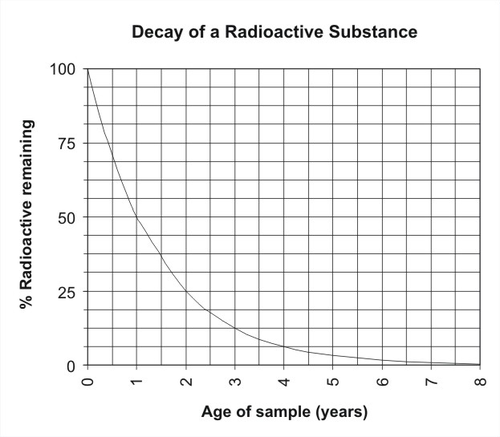
Calculating Half-life
A radioactive half-life refers to the amount of time it takes for half of the original isotope to decay. For example, if the half-life of a 50.0 gram sample is 3 years, then in 3 years only 25 grams would remain. During the next 3 years, 12.5 grams would remain and so on.
Use the How to Calculate Half-Life .pdf as a step-by-step for calculating half-life. The T-chart method is how we will calculate half-life in class.
Use the How to Calculate Half-Life .pdf as a step-by-step for calculating half-life. The T-chart method is how we will calculate half-life in class.

Carbon-14 Dating
Earth's atmosphere contains three isotopes of carbon. Carbon-12 is stable and accounts for 98.9% of atmospheric carbon. Carbon-13 is also stable and accounts for 1.1% of atmospheric carbon. Carbon-14 is radioactive and is found in tiny amounts. Carbon-14 is produced naturally in the atmosphere when cosmic rays interact with nitrogen atoms. The amount of carbon-14 produced in the atmosphere at any particular time has been relatively stable through time.
Radioactive carbon-14 decays to stable nitrogen-14 by releasing a beta particle. The nitrogen atoms are lost to the atmosphere, but the amount of carbon-14 decay can be estimated by measuring the proportion of radioactive carbon-14 to stable carbon-12. As a substance ages, the relative amount of carbon-14 decreases.
Carbon is removed from the atmosphere by plants during the process of photosynthesis. Animals consume this carbon when they eat plants or other animals that have eaten plants. Therefore carbon-14 dating can be used to date plant and animal remains. Examples include timbers from an old building, bones, or ashes from a fire pit. Carbon dating can be effectively used to find the age of materials between 100 and 50,000 years old.
Earth's atmosphere contains three isotopes of carbon. Carbon-12 is stable and accounts for 98.9% of atmospheric carbon. Carbon-13 is also stable and accounts for 1.1% of atmospheric carbon. Carbon-14 is radioactive and is found in tiny amounts. Carbon-14 is produced naturally in the atmosphere when cosmic rays interact with nitrogen atoms. The amount of carbon-14 produced in the atmosphere at any particular time has been relatively stable through time.
Radioactive carbon-14 decays to stable nitrogen-14 by releasing a beta particle. The nitrogen atoms are lost to the atmosphere, but the amount of carbon-14 decay can be estimated by measuring the proportion of radioactive carbon-14 to stable carbon-12. As a substance ages, the relative amount of carbon-14 decreases.
Carbon is removed from the atmosphere by plants during the process of photosynthesis. Animals consume this carbon when they eat plants or other animals that have eaten plants. Therefore carbon-14 dating can be used to date plant and animal remains. Examples include timbers from an old building, bones, or ashes from a fire pit. Carbon dating can be effectively used to find the age of materials between 100 and 50,000 years old.
Applications, Benefits, and Problems of Nuclear Energy
Watch the following video making note in your Interactive Notebook of the applications, benefits, and problems of Nuclear Energy. Which side do you stand on??? Are you pro- or con- towards nuclear energy?
The Eyes of Nye- Nuclear Energy
The Eyes of Nye- Nuclear Energy
Nuclear Energy Explained: How does it work?